Inari Clottriever Brochure
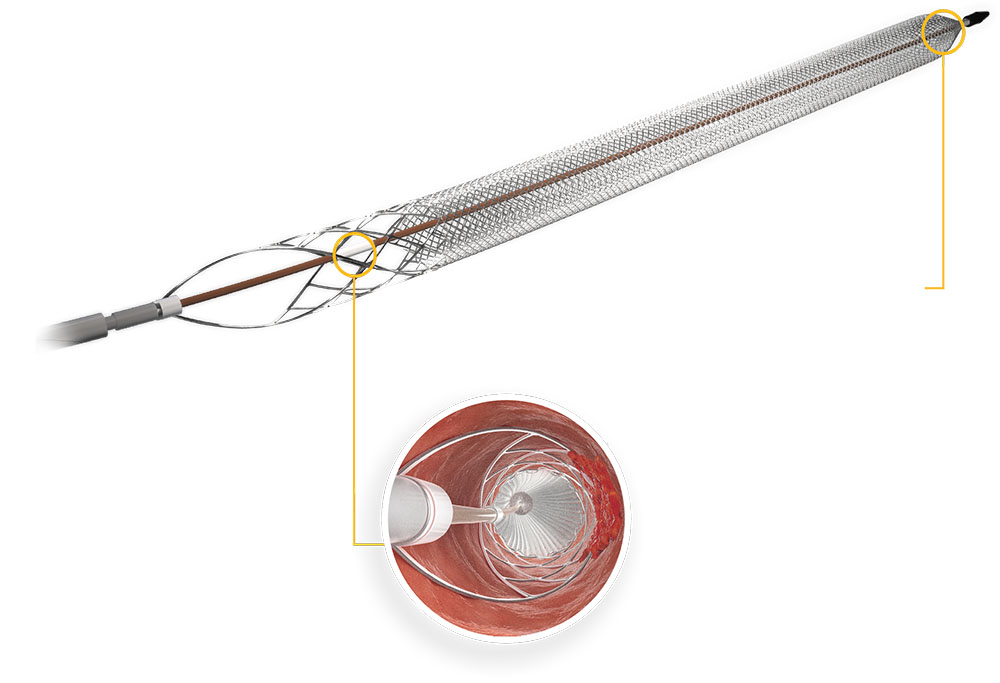
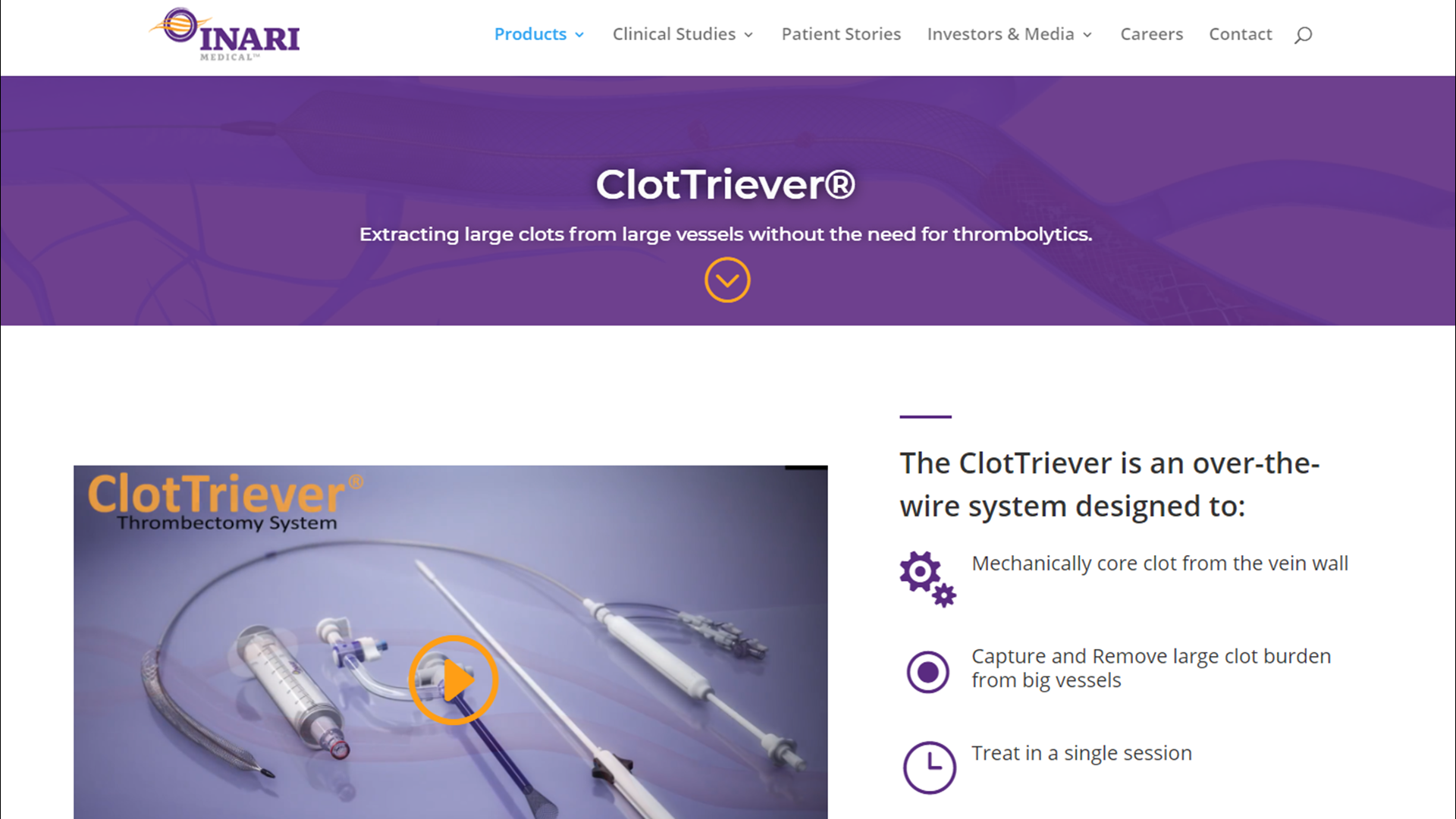
Inari Clottriever Brochure - • mechanically core clot from vein wall • capture and remove large clot burden from big vessels • treat in a single session • eliminate. Fda 510(k) clearance received for peripheral thrombectomy by inari | apr 10, 2017 | news The clottriever® catheter is a purely mechanical treatment option that liberates thrombus from the vessel walls and captures in a collection bag to be extracted via the clottriever® sheath. The clottriever® is a mechanical thrombectomy system designed to remove large clots from large vessels to treat deep vein thrombosis (dvt) in a single session, without the need for. Inari medical announces first patient treated with the clottriever™ thrombectomy system; As the first mechanical thrombectomy system to receive fda 510 (k) clearance for the treatment of pulmonary embolism, the flowtriever system is designed for rapid thrombus removal and. In addition, it is designed to give physicians advanced control in chronic. The clottreiver® system by inari medical® is available to treat the full range of acute and chronic deep vein thrombosis. The inari clottriever is a percutaneous mechanical thrombectomy system consisting of an expandable nitinol collection bag that is dragged along the vein wall,. The clottriever thrombectomy system is indicated for: The clottriever thrombectomy system is intended for use in the peripheral vasculature including deep vein thrombosis (dvt). The clottriever® system is a medical device fda cleared for the treatment of deep vein thrombosis. The clottriever thrombectomy system is indicated for: The clottriever® catheter is a purely mechanical treatment option that liberates thrombus from the vessel walls and captures in a collection bag to be extracted via the clottriever® sheath. Fda 510(k) clearance received for peripheral thrombectomy by inari | apr 10, 2017 | news As the first mechanical thrombectomy system to receive fda 510 (k) clearance for the treatment of pulmonary embolism, the flowtriever system is designed for rapid thrombus removal and. See instructions for use for complete indications for use,. 2) 2) injection, infusion, and/or aspiration of. In addition, it is designed to give physicians advanced control in chronic. The clottriever® is a mechanical thrombectomy system designed to remove large clots from large vessels to treat deep vein thrombosis (dvt) in a single session, without the need for. The inari clottriever is a percutaneous mechanical thrombectomy system consisting of an expandable nitinol collection bag that is dragged along the vein wall,. Inari medical announces first patient treated with the clottriever™ thrombectomy system; 2) 2) injection, infusion, and/or aspiration of. The clottriever thrombectomy system is indicated for: The clottreiver® system by inari medical® is available to treat the full. The clottriever® system is a medical device fda cleared for the treatment of deep vein thrombosis. The clottreiver® system by inari medical® is available to treat the full range of acute and chronic deep vein thrombosis. The clottriever® system • designed to core, capture and remove large clots from large venous vessels • in february of 2017, the clottriever system. See instructions for use for complete indications for use,. Fda 510(k) clearance received for peripheral thrombectomy by inari | apr 10, 2017 | news The clottriever® catheter is a purely mechanical treatment option that liberates thrombus from the vessel walls and captures in a collection bag to be extracted via the clottriever® sheath. In addition, it is designed to give. As the first mechanical thrombectomy system to receive fda 510 (k) clearance for the treatment of pulmonary embolism, the flowtriever system is designed for rapid thrombus removal and. The clottriever® system • designed to core, capture and remove large clots from large venous vessels • in february of 2017, the clottriever system received 510(k). See instructions for use for complete. The clottriever thrombectomy system is intended for use in the peripheral vasculature including deep vein thrombosis (dvt). 2) 2) injection, infusion, and/or aspiration of. The clottriever® system is a medical device fda cleared for the treatment of deep vein thrombosis. The clottriever® is a mechanical thrombectomy system designed to remove large clots from large vessels to treat deep vein thrombosis. See instructions for use for complete indications for use,. The clottriever® catheter is a purely mechanical treatment option that liberates thrombus from the vessel walls and captures in a collection bag to be extracted via the clottriever® sheath. The clottriever thrombectomy system is indicated for: The clottriever® system • designed to core, capture and remove large clots from large venous. The clottriever® is a mechanical thrombectomy system designed to remove large clots from large vessels to treat deep vein thrombosis (dvt) in a single session, without the need for. The clottriever thrombectomy system is indicated for: The clottriever thrombectomy system is intended for use in the peripheral vasculature including deep vein thrombosis (dvt). In addition, it is designed to give. The clottriever® system is a medical device fda cleared for the treatment of deep vein thrombosis. The clottreiver® system by inari medical® is available to treat the full range of acute and chronic deep vein thrombosis. The clottriever® catheter is a purely mechanical treatment option that liberates thrombus from the vessel walls and captures in a collection bag to be. Inari medical announces first patient treated with the clottriever™ thrombectomy system; The clottriever® system • designed to core, capture and remove large clots from large venous vessels • in february of 2017, the clottriever system received 510(k). The clottriever thrombectomy system is indicated for: Fda 510(k) clearance received for peripheral thrombectomy by inari | apr 10, 2017 | news The. The inari clottriever is a percutaneous mechanical thrombectomy system consisting of an expandable nitinol collection bag that is dragged along the vein wall,. The clottriever® system • designed to core, capture and remove large clots from large venous vessels • in february of 2017, the clottriever system received 510(k). 2) 2) injection, infusion, and/or aspiration of. In addition, it is. The clottriever thrombectomy system is indicated for: Inari medical announces first patient treated with the clottriever™ thrombectomy system; The clottriever® is a mechanical thrombectomy system designed to remove large clots from large vessels to treat deep vein thrombosis (dvt) in a single session, without the need for. 2) 2) injection, infusion, and/or aspiration of. As the first mechanical thrombectomy system to receive fda 510 (k) clearance for the treatment of pulmonary embolism, the flowtriever system is designed for rapid thrombus removal and. The clottriever® catheter is a purely mechanical treatment option that liberates thrombus from the vessel walls and captures in a collection bag to be extracted via the clottriever® sheath. The clottreiver® system by inari medical® is available to treat the full range of acute and chronic deep vein thrombosis. • mechanically core clot from vein wall • capture and remove large clot burden from big vessels • treat in a single session • eliminate. The clottriever® system is a medical device fda cleared for the treatment of deep vein thrombosis. The clottriever thrombectomy system is intended for use in the peripheral vasculature including deep vein thrombosis (dvt). The clottriever® system • designed to core, capture and remove large clots from large venous vessels • in february of 2017, the clottriever system received 510(k). The inari clottriever is a percutaneous mechanical thrombectomy system consisting of an expandable nitinol collection bag that is dragged along the vein wall,.ClotTriever system for removing large clots from large vessels and
(PDF) Percutaneous Thrombectomy of Upper Extremity and Thoracic Central
ClotTriever system for removing large clots from large vessels and
(PDF) Percutaneous Thrombectomy of Upper Extremity and Thoracic Central
Inari Medical ClotTriever® Healthcare Essentials
Inari Medical VIR ClotTriever November 18, 2020
Inari Medical enrolls first patient in ClotTriever trial
Inari 40102 ClotTriever Catheter 11Fr
Inari Medical ClotTriever® Healthcare Essentials
(PDF) Successful removal of an acute deep vein thrombosis by the INARI
Fda 510(K) Clearance Received For Peripheral Thrombectomy By Inari | Apr 10, 2017 | News
In Addition, It Is Designed To Give Physicians Advanced Control In Chronic.
See Instructions For Use For Complete Indications For Use,.
Related Post: